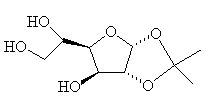
Chinese Factory Supply Wholesale Monoacetone-D-Glucose 18549-40-1 with Cheap Price
Synonyms: Acetone Glucose, Acetone-D-Glucose, 1,2-O-Isopropylidene-α-D-glucofuranose, 1,2-O-Isopropylidene-alpha-D-glucofuranose, 1,2-O-(1-methylethylidene)-α-D-glucofuranose, Monoacetone Glucofuranose.
Molecular Formula: C9H16O6
Molecular weight: 220.22
CAS No.: 18549-40-1
EC No.: 242-420-9
Quality Standard:
|
Item
|
Specification
|
|
Appearance
Assay (GC), %
Melting point, ℃
Specific rotation, 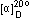 (C=2,H2O) (C=2,H2O)
Loss on drying, %
Residue on ignition, %
Heavy metals, ppm
TLC
|
White microcrystalline solid
≥98
158~162
-10~ -14
≤1.0
≤0.2
≤10
Single spot
|
Properties: It is white microcrystalline solid. Dissolve in water, acetone, ethanol, tetrahydrofuran, dimethyl formamide etc.
1,2-O-Isopropylidene-D-glucofuranose(Cas 18549-40-1) Usage
This product is an important pharmaceutical intermediates. Used in synthesis Tribenoside,synthesis nojirimycin and deoxynojirimycin for treatment diabetes, complexes that carbohydrate combine with organic metal including platinum, osmium tetroxide, ruthenium, etc for treatment cancer, other anticancer or antitumor pharmaceuticals and their precursor such as organic phosphide, and azole that contain glycosyl for resistance to junin virus and fever virus such as dengue fever, dengue hemorrhagic fever and Argentine hemorrhagic fever.
Storage: Store in a tightly closed container.Store in a cool,dry area.
Package: 25kgs/drum or as required by the customer.
Expiration Date: 2 years.
18549-40-1 Relevant articles
Polymer-supported ferric chloride as a heterogeneous catalyst for chemoselective deprotection of acetonides
Chari, M. Adharvana,Syamasundar
, p. 708 - 710 (2005)
Acetonides undergo chemoselective deprot...
Thermal and Lewis acid promoted intramolecular Diels-Alder reaction of furanose tethered 1,3,9-decatriene systems: A synthetic and computational investigation
Kalmode, Hanuman P.,Maity, Dilip K.,Bhate, Prakash M.
, p. 63445 - 63462 (2016)
The intramolecular Diels-Alder (IMDA) re...
-
Seebeck et al.
, p. 1142,1145 (1944)
-
Hydrogen isotopic profile in the characterization of sugars. Influence of the metabolic pathway
Zhang, Ben-li,Billault, Isabelle,Li, Xiaobao,Mabon, Francoise,Remaud, Gerald,Martin, Maryvonne L.
, p. 1574 - 1580 (2002)
The site-specific natural hydrogen isoto...
Removal of O-benzyl protecting-groups of carbohydrate derivatives by catalytic, transfer hydrogenation
Rao, Vanga S.,Perlin, Arthur S.
, p. 175 - 177 (1980)
-
-
Roseman
, p. 4467 (1952)
-
-
Williams,Loewus
, p. 149,150-155 (1978)
-
Chiron approach for the synthesis of (1S,2R,5R,7S)-2-hydroxy-exo-brevicomin
Gautam,Kumar, D. Naveen,Rao, B. Venkateswara
, p. 819 - 821 (2006)
(1S,2R,5R,7S)-2-Hydroxy-exo-brevicomin e...
Quantification of deuterium isotopomers of tree-ring cellulose using nuclear magnetic resonance
Betson, Tatiana R.,Augusti, Angela,Schleucher, Juergen
, p. 8406 - 8411 (2006)
Stable isotopes in tree rings are import...
-
Buchanan,Oakes
, p. 242,247 (1965)
-
INTERACTION BETWEEN ACETONE AND SOME CARBOHYDRATE BENZENEBORONATES: SELECTIVE ACETONOLYSIS OF 2-PHENYL-1,3,2-DIOXABOROLANES
Griffiths, Chridtopher J.,Weigel, Helmut
, p. 17 - 22 (1980)
Treatment of D-glucitol 1,3:2,4:5,6-tris...
-
Mehltretter et al.
, p. 2424,2426 (1951)
-
A Warburg effect targeting vector designed to increase the uptake of compounds by cancer cells demonstrates glucose and hypoxia dependent uptake
Glenister, Alexandra,Simone, Michela I.,Hambley, Trevor W.
, (2019)
Glycoconjugation to target the Warburg e...
A mild and convenient approach for selective acetonide cleavage involved in carbohydrate synthesis using PPA-SiO2
Nikam, Rahul R.,Gore, Kiran R.
, p. 63 - 74 (2020)
Here, we report a highly selective, effi...
Acetalation studies. Part IX. Reaction of sucrose and some related sugars with acetone in the presence of iodine; a novel cleavage-isopropylidenation method
Verhart, Cor G. J.,Caris, Brigitte M. G.,Zwanenburg, Binne,Chittenden, Gordon J. F.
, p. 348 - 352 (1992)
An efficient cleavage-isopropylidenation...
A mild and efficient procedure to remove acetal and dithioacetal protecting groups in carbohydrate derivatives using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone
Garcia Fernandez,Ortiz Mellet,Moreno Marin,Fuentes
, p. 263 - 268 (1995)
-
-
Kiso,Hasegawa
, p. 95,96-101 (1976)
-
Synthesis and antimicrobial activity of (?)-cleistenolide and analogues
Benedekovi?, Goran,Popsavin, Mirjana,Radulovi?, Niko S.,Stojanovi?-Radi?, Zorica,Farkas, Sándor,Francuz, Jovana,Popsavin, Velimir
, (2020/12/07)
Using the “chiral pool” approach, two mo...
Nascent-HBr-Catalyzed Removal of Orthogonal Protecting Groups in Aqueous Surfactants
Bera, Smritilekha,Gupta, Shilpi,Mondal, Dhananjoy
, (2020/02/04)
Organic reactions in the aqueous environ...
Diastereoisomerically pure, (S)-O-1,2-O-isopropyli dene-(5-O-α-d-glucofuranosyl) t-butanesulfinate: Synthesis, crystal structure, absolute configuration and reactivity
B?aszczyk, Jaros?aw,Bujnicki, Bogdan,Chmielewski, Marek,Drabowicz, Józef
, (2020/09/16)
The reaction of t-butylmagnesium chlorid...
18549-40-1 Process route
-
- 20880-92-6
2,3;4,5-di-O-isopropylidene-β-D-fructopyranose
-
- 66900-93-4
1,2-O-isopropylidene-β-D-fructopyranose
-
- 18549-40-1
1,2-O-isopropylidene-α-D-glucofuranose
Conditions
| Conditions |
Yield |
|
With acetic acid; iodine; Yield given. Multistep reaction. Yields of byproduct given; 1) reflux, 4 h, 2) room temp., 22 h;
|
|
|
With acetic acid; iodine; Yield given. Multistep reaction. Yields of byproduct given; 1) reflux, 4 h, 2) room temp., 22 h;
|
|
-
- 20880-92-6
2,3;4,5-di-O-isopropylidene-β-D-fructopyranose
-
- 18549-40-1
1,2-O-isopropylidene-α-D-glucofuranose
Conditions
| Conditions |
Yield |
|
acetone; Sucrose; With phosphoric acid; zinc(II) chloride;
With acetic acid;
|
|
18549-40-1 Upstream products
-
50-99-7
D-glucose
-
67-64-1
acetone
-
582-52-5
1,2:5,6-di-O-isopropylidene-α-D-glucofuranose
-
3067-56-9
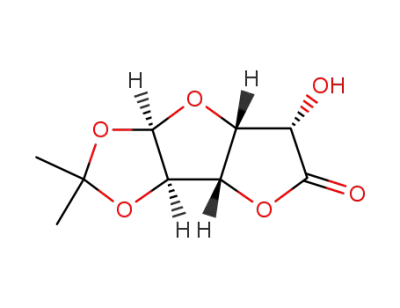
D-glucurono-3,6-lactone acetonide
18549-40-1 Downstream products
-
56-73-5
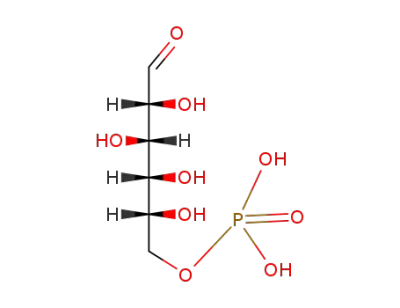
D-glucose 6-phosphate
-
29364-56-5
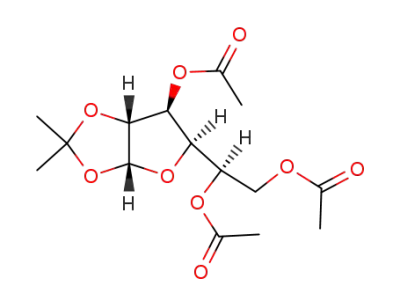
3,5,6-tri-O-acetyl-1,2-O-isopropylidene-α-D-glucofuranose
-
33737-08-5
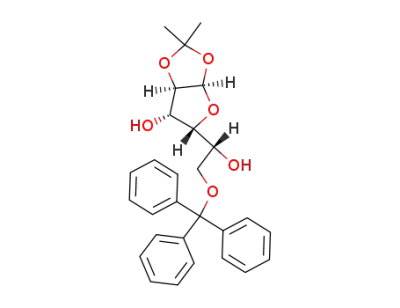
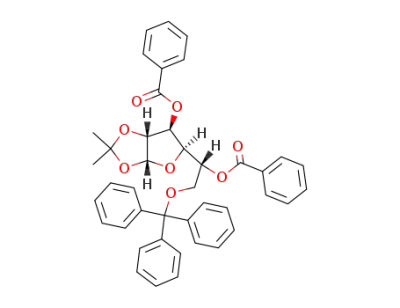
1,2-O-isopropylidene-6-O-trityl-α-D-glucofuranoside
-
64429-78-3
O3,O5-dibenzoyl-O1,O2-isopropylidene-O6-trityl-α-D-glucofuranose